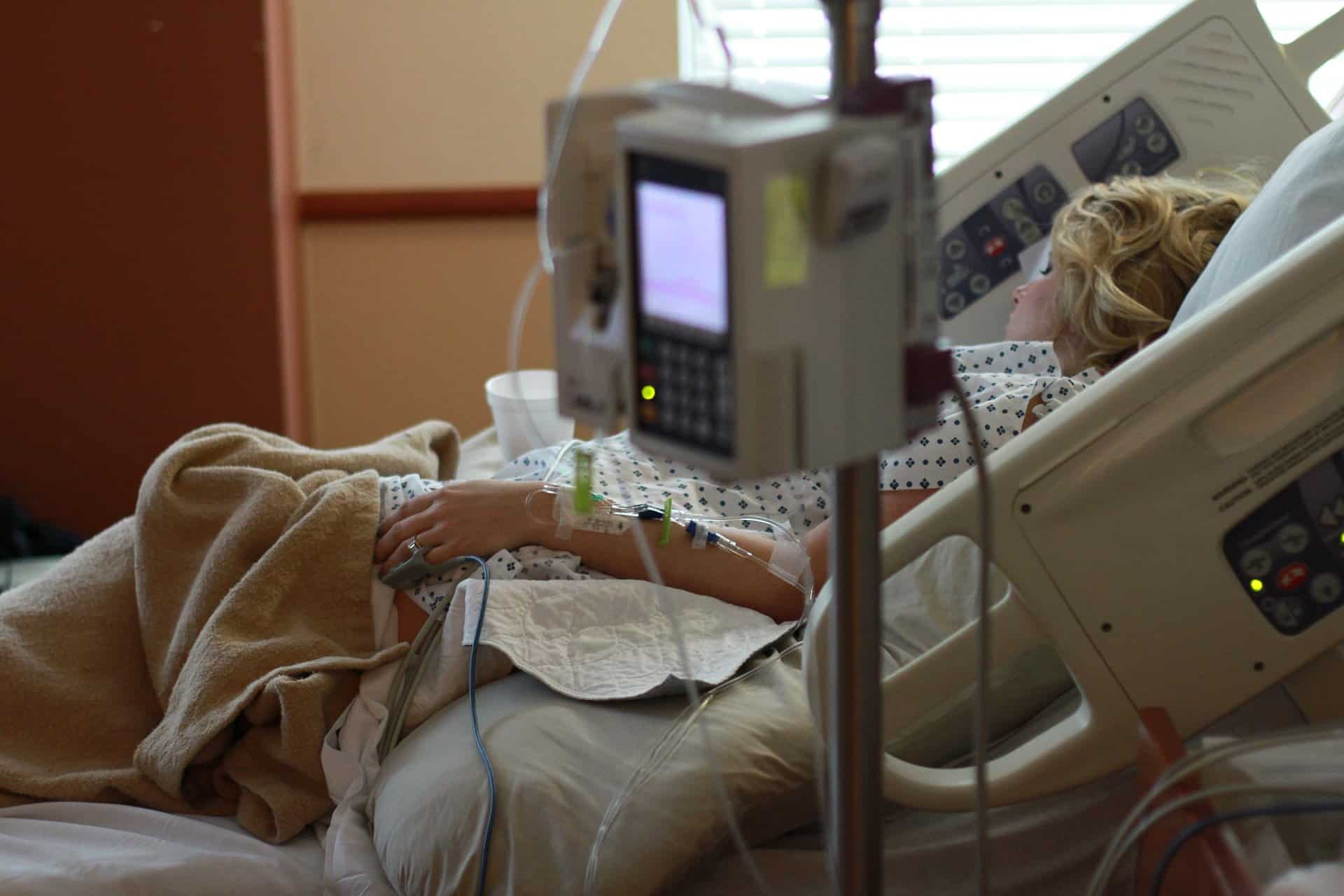
Covid-19 treatments using plasma taken from the blood of recovered coronavirus patients should not be given to people with mild or moderate illness, the World Health Organization said on Tuesday, December 7.
Convalescent plasma showed some early promise when given intravenously to people sick with Covid-19.
But in advice published in the British Medical Journal, the WHO now says that “current evidence shows that it does not improve survival nor reduce the need for mechanical ventilation, and it is costly and time-consuming to administer”.
It made a “strong recommendation” against the use of blood plasma in people who do not have serious Covid-19 symptoms, and said that even for patients with severe and critical illness, the treatment should only be given as part of a clinical trial.
Convalescent plasma is the liquid part of blood from a recovered Covid-19 patient that contains antibodies produced by the body after being infected.
It was one in an array of potential treatments investigated early in the pandemic, but has shown limited benefit in clinical trials.
The WHO said its latest recommendations were based on evidence from 16 trials involving 16,236 patients with non-severe, severe, and critical Covid-19 infection.